Dear Doctor Letter
Dear Doctor:
We're writing to urge you to review the abundance of biological evidence linking induced abortion with increased breast cancer risk, so that you can consider what legal obligations you might have to properly inform your patients.
General counsel for a medical organization informed doctors in 2005 that women from Pennsylvania, Oregon and Australia have successfully sued their abortion providers for neglecting to warn them about the increased risks of breast cancer and emotional harm. 1
A Ninth Circuit Court decision on the law of informed consent highlights a doctor's duty to inform his or her patients about the available evidence: "[W]e believe a risk must be disclosed even if it is but a potential risk rather than a conclusively determined risk. . . . Medical knowledge should not be limited to what is generally accepted as a fact by the profession.... To justify ignorance of this type of risk would insulate the medical profession beyond what is legally acceptable. . . . A literature search will thus put a physician on notice of these risks." 2
A British researcher presented his findings on "Trends and Risk Factors in English Breast Cancer" at the British Cancer Research Meeting 2004 in Manchester. 3 He found that legally induced abortion is the "best predictor" of English breast cancer trends. Because he relied on historic national data reporting breast cancers and abortions in England and Wales, his research is free of any possibility of recall bias. He described nulliparous abortions as "highly carcinogenic." His findings agree with what is known about human biology, endocrinology and oncology.
Abortion was cited as a risk factor in the 1991 and 1997 editions of an authoritative medical text used by breast disease specialists. 4 The authors cited a 1988 special lecture, "Estrogens as a Cause of Human Cancer: The Richard and Linda Rosenthal Foundation Award Lecture." 5
Estrogen is a mitogen and a carcinogen. It stimulates proliferation and promotes the growth of cancer. Estrogen also acts a cancer initiator. Mutations can occur during proliferation, resulting in a malignancy. Scientists added steroidal estrogens (used in hormone replacement therapy and oral contraceptives) to the nation's list of carcinogens in 2002. 6
David Longfellow, Ph.D., chief of the Chemical and Physical Carcinogenesis Branch at the National Cancer Institute, reported that "It has been an uphill battle to convince the mainstream that estrogen initiates cancer by damaging DNA." 7 Human breast epithelial cells without estrogen receptors became cancerous and had genetic damage after being exposed to estrogen. Some estrogen metabolites directly damage DNA, i.e. free radicals and CE-quinone. Estrogen is metabolized in four steps to CE-quinone. CE-quinone pulls purine bases out of DNA strands, directly damaging DNA. Rogan reported that women with breast cancer have higher levels of CE-quinone than do women without the disease. 8
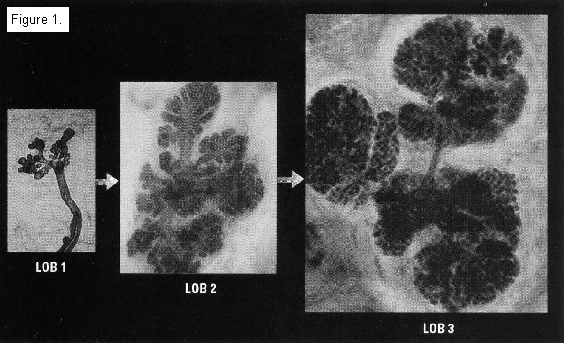
Russo and Russo's pioneering research on the connection between breast tissue maturity and the development of breast cancer identified four types of breast lobules. 9 10 11 Type 1 lobules, the most primitive and undifferentiated, are present before menarche. After menarche, some breast tissue evolves into Type 2 lobules. These are more complex and include more ductules per lobule.
The most cancer-resistant tissue, types 3 and 4 breast lobules, only develops during the third trimester of pregnancy. During the first two trimesters, estrogen stimulates cell proliferation and lobule formation. However, starting at 32 weeks gestation, there is differentiation of breast tissue, resulting in a decrease in the number of terminal end buds, increased lobular development, and a termination of the proliferative process. Type 3 breast lobules average 80 ductules or alveoli per lobule. Type 4 lobules are milk-producing. This accounts for the fact that women who have premature deliveries before 32 weeks double their breast cancer risk. 12 13
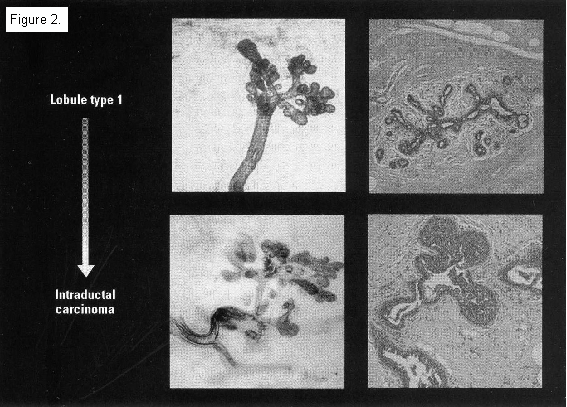
Type 1 and 2 lobules grow faster in vitro than Type 3 lobules, have a shorter doubling time than Type 4, and have higher DNA labeling indicies than do Types 3 and 4 lobules. Type 1 lobules are terminal ductal lobular units. This is where ductal cancers develop. Parous women with breast cancer have a greater percentage of Type 1 lobules than do parous women without the disease.
Women have the greatest number of Types 1 and 2 lobules and are the most vulnerable to carcinogens during the time between menarche and the birth of a first child. Due to the proliferation of lobules during the first two trimesters, the woman who has an induced abortion is left with more Types 1 and 2 lobules than she had before her pregnancy began. This leaves her with more places for cancer to start. By contrast, the woman who has a full term pregnancy is left with more mature, cancer-resistant Types 3 and 4 lobules than she had before her pregnancy began. This results in the protective effect of a full term pregnancy.
No scientific team currently claims to have found credible evidence of recall bias. However, it's far more logical to hypothesize that women who don't want to reveal their abortion histories would choose not to participate in an abortion-breast cancer study in the first place. It makes little sense to argue that women would agree to participate and then decide to lie about their abortion histories. See the "Bias" page on our Web site for a discussion of this more logical alternative to recall bias and how it might impact scientists' findings.
For the sake of your patients, we urge you to familiarize yourself with the evidence. For additional information about the research, please see these web sites:
www.AbortionBreastCancer.com and www.BCPInstitute.org.
Sincerely yours,
Karen Malec
President
During a normal pregnancy, women are overexposed to the hormone estrogen (a recognized carcinogen). Estrogen causes breast tissue to grow. It stimulates Types 1 and 2 lobules to multiply. Abortion leaves a woman with more Types 1 and 2 lobules.
Only a third trimester process in pregnancy protects a woman from estrogen overexposure and matures her breast tissue into cancer-resistant, milk producing breast tissue (Types 3 & 4 lobules). A full term pregnancy leaves a woman with an increase in cancer-resistant breast lobules. That's why women with more children have a lower lifetime risk for the disease.
A woman is the most vulnerable to carcinogens between the time that her menstrual periods begin and her first full term pregnancy occurs. With every menstrual period, estrogen stimulates Types 1 and 2 lobules.
*Whole mount preparation, toluidine blue stain (Lob 1, 2 and 3: x99, x118 and x121, respectively).
+Whole mounts stained with toluidine blue, histological sections with hematoxylin and eosin.
- 1. Schlafly A. Legal implications of a link between abortion and breast cancer. J Am Phys Surgeons 2005;10:11-14.
- 2. Harbeson v. Parke Davis, Inc., 746 F.2d 517, 525 (9th Cir. 1984).
- 3. Carroll P. Br J Cancer. Vol. 91, Supplement 1, July 2004, page S24.
- 4. Bland, KI, Copeland, EM, The Breast: Comprehensive Management of Benign and Malignant Diseases, 2nd ed, Saunders, Phila, NY 1997; v. 1:523.
- 5. Henderson BE, et al. Cancer Research 48, 246-253, January 15, 1988.
- 6. "Report on Carcinogens," U.S. National Toxicology Program, December 11, 2002.
- 7. Miller K. Jrnl Natl Cancer Inst, Vol 95, No. 2, 100-102, January 15, 2003.
- 8. Rogan, Ph.D., University of Nebraska, San Antonio Breast Cancer Symposium 2002.
- 9. Russo J, et al. Cancer Res 1988;48:2837-2857
- 10. Russo J, et al. Breast Cancer Research and Treatment (1992)23:211-218.
- 11. Russo J, et al. J Natl Cancer Inst Monogr 2000; 27:17-37.
- 12. Hsieh et al. Lancet 1999;353:1239.
- 13. Melbye et al. Br J Cancer 1999;80:609-12.